1 min read
This Is What an Efficient Marketing Workflow Actually Looks Like
Most marketing teams are busier than ever. Campaigns are running across more channels, content volumes are increasing, and the pressure to deliver...
12 min read
 Rebecca Freeman
:
April 28, 2026
Rebecca Freeman
:
April 28, 2026

Pharma marketing teams live with a tension that never quite goes away. The pressure to launch campaigns quickly is real. So is the obligation to get every word, claim, and image signed off by legal, regulatory, and medical reviewers before anything goes out the door.
When those two forces collide, something usually gives. Either campaigns slip their launch dates while waiting for approvals, or shortcuts creep in and compliance suffers. Neither outcome is acceptable in a regulated environment where a missed review step can trigger regulatory action, damage a brand, or put patients at risk.
Our guide is for pharma marketing professionals, compliance officers, brand managers, and regulatory teams who want to understand where approval processes break down, what high-performing teams do differently, and how the right tools and workflows can help you move faster without cutting corners.
In most industries, marketing content goes through a relatively straightforward sign-off process. A few people review it, give feedback, and it gets published.
In pharma, that process involves multiple layers of scrutiny, each with its own requirements.
Multiple stakeholders with different priorities. A piece of promotional material might need sign-off from marketing, brand, medical affairs, regulatory affairs, legal, and compliance. Each function has a different lens. Marketing wants compelling copy. Medical wants accuracy. Regulatory wants every claim to be substantiated. Legal wants to avoid liability. Aligning these perspectives takes time and careful coordination.
Strict regulatory requirements. Depending on where a company operates, promotional content must comply with guidelines from bodies such as the MHRA, FDA, EMA, and PMCPA, as well as internal standard operating procedures (SOPs) that govern how claims are made and substantiated. Getting any of this wrong carries significant consequences.
High stakes for errors. A factual inaccuracy, an unsubstantiated claim, or a missing safety statement isn't just embarrassing. It can lead to regulatory warnings, mandatory recalls of materials, fines, or reputational damage that takes years to repair.
Detailed review processes. Because of the above, pharma organizations typically require multiple rounds of review and formal sign-off from qualified individuals. These processes exist for good reason. But without the right infrastructure to support them, they slow everything down.
A pharma marketing approval workflow is the structured process through which promotional content moves from creation to final sign-off before publication or distribution.
It typically includes:
The approval workflow sits at the heart of the wider marketing content process. It connects creative output to compliance requirements, and when it works well, it acts as a quality assurance mechanism rather than a bottleneck.
The challenge is that many organizations have the right intentions but the wrong infrastructure. Workflows exist in theory but break down in practice.

Understanding where things go wrong is the first step to fixing them.
Bringing in more reviewers at the wrong time, or involving people who don't have a clear role in the process, creates confusion about who has decision-making authority. When ten people can comment but only two can approve, feedback accumulates without resolution.
Without clear ownership at each stage, reviews stall. Reviewers wait for each other. Nobody knows whether a piece is approved, in review, or back with the creator for revisions.
Email is still the backbone of many pharma approval workflows, and it creates serious problems. Documents get attached and re-attached. Reviewer feedback arrives at different times and in different formats. Tracking which version is current becomes a project in itself.
Manual processes are also difficult to audit. When a regulator asks for evidence of who reviewed what and when, email chains are an unreliable paper trail.
When reviewers use different tools to annotate, comment, or track changes, consolidating feedback becomes a major task in its own right. Marketing teams end up spending hours reconciling comments from Word documents, PDFs, emails, and verbal feedback before they can even begin a revision.
This scatter also makes it easy for conflicting instructions to slip through. Two reviewers may give contradictory direction on the same piece of copy, and without a centralized view, there's no easy way to resolve it.
Version proliferation is one of the most common and most damaging problems in creative approval workflows. When documents are shared by email or stored in personal drives, it's easy to end up with multiple versions of the same file circulating at the same time.
Reviewers can find themselves providing feedback on an outdated draft. Final approvals get attached to the wrong version. Teams make changes that have already been rejected. The result is wasted effort, confusion, and real compliance risk.
Regulated industries require clear records of the approval process. Who reviewed a document? What feedback did they give? When was it approved and by whom? What version was signed off?
Without a system that captures this automatically, teams have to reconstruct audit trails from memory and scattered documentation. That's time-consuming, error-prone, and often inadequate when a formal review is required.
Even when stakeholders are engaged and processes are clear, review cycles can drag simply because there's no urgency mechanism built in. Reviewers have full diaries. Without clear deadlines, a clear escalation path, and visibility into where a piece sits in the workflow, reviews default to "when I have time."
In a sector where campaign timing can coincide with clinical trial milestones, product launches, or regulatory windows, slow reviews have real commercial consequences.
The true cost of a broken approval process goes beyond frustration.
Delayed campaign launches. When approvals take longer than planned, launch dates slip. In competitive therapeutic areas, being late to market with patient education materials or HCP communications is a genuine disadvantage.
Increased compliance risk. The more informal and undocumented the approval process, the greater the risk that something slips through that shouldn't. Gaps in oversight, missed review stages, or approvals given verbally rather than in writing all increase exposure.
Higher operational costs. Rework is expensive. When version control fails and approved content is edited again, or when teams have to rebuild audit trails from scratch, the hours add up quickly. These are costs that could be eliminated with better process infrastructure.
Frustrated teams. Marketing and agency teams that spend more time chasing approvals than creating content aren't operating at full potential. Reviewer fatigue also sets in when the process is disorganized. People stop engaging carefully when they're bombarded with unclear requests.
Lost market opportunities. Timely, compliant communication is a competitive advantage. Organizations that can move efficiently from creation to approved content will consistently outperform those still working through backlogged reviews.

Teams that manage approvals well don't just have better tools. They have better processes, clearer roles, and a shared understanding of how the workflow is supposed to function.
High-performing teams map their approval process into defined stages, each with a clear purpose, assigned reviewers, and an expected timeframe. Rather than routing every document to every stakeholder simultaneously, they build sequential or parallel review pathways appropriate to the type of content.
For example, a promotional leavepiece might go through a medical accuracy review before it reaches regulatory, ensuring that fundamental scientific claims are correct before compliance language is assessed. A patient support material might follow a different track entirely.
Every stage of the approval process should have a named owner. Someone is responsible for initiating the review, someone is responsible for each sign-off, and someone has authority to close out conflicting feedback. Without this clarity, approvals stall at every stage.
High-performing teams often document roles in their SOPs and ensure that everyone involved in the process knows their responsibilities before a project starts, not after it has already been delayed.
Consolidating all feedback in a single environment eliminates the confusion that comes from scattered comments across tools and email threads. When every reviewer works within the same system, conflicts are visible, revisions are tracked, and the current version is always clear.
This is where purpose-built platforms provide significant value over general document management or email-based workflows. Centralized collaboration isn't just more convenient. It's materially safer.
Teams that can see exactly where a piece of content is in the approval cycle, who has reviewed it, what comments are outstanding, and what has been resolved are far better positioned to manage timelines and anticipate delays before they become problems.
Dashboards and status views that surface this information in real time reduce the need for chasing emails and status meetings, and give project managers the data they need to make decisions quickly.
Automatically capturing a complete record of every review action, comment, approval, and rejection is essential in regulated environments. When audit trails are built into the workflow system, compliance teams don't have to construct them manually. They exist by default.
This also supports continuous improvement. Being able to review where approvals slowed down, which stages generated the most revision cycles, or which types of content took longest to approve gives teams the data to refine their processes over time.
Approval speed improves when reviewers have the right context, clear instructions, and a clean working environment. Teams that provide reviewers with a clear brief, the full content in a single consolidated view, and a structured mechanism for giving feedback typically get faster and better quality responses.
Setting and communicating review deadlines, with built-in reminders and escalation paths, also keeps cycles moving without requiring constant manual follow-up.
Automation is often discussed as a way to speed things up, but in regulated marketing environments, its primary value is consistency.
When approval workflows are automated, every piece of content follows the same path. The right reviewers are notified at the right time. Deadlines are enforced. Documents can't be moved to the next stage until the current stage is complete. This structural consistency reduces the likelihood of a review step being skipped or a version being approved in error.
Automation also reduces the administrative burden on marketing and project teams. Chasing reviewers, tracking versions, and compiling audit records are tasks that automation handles in the background, freeing teams to focus on content quality.
For compliance officers, automated workflows provide a layer of assurance that manual processes can't. When they can see that every document followed the defined path, every required reviewer signed off, and every approval was captured in the system, they can validate compliance with confidence.
Importantly, automation doesn't mean removing human judgment from the process. It means ensuring that human judgment is applied at the right points, with the right information, every time.

Online proofing is a component of the wider approval workflow that deserves specific attention in pharma marketing.
In traditional review processes, reviewers annotate PDFs, make comments in Word documents, or provide verbal or written feedback by email. This approach is fragile. It's difficult to track what changed, who said what, and whether a revision addressed the original comment.
Online proofing platforms replace this with a structured, digital review environment where reviewers can annotate content directly, see each other's comments, and track whether issues have been resolved. For pharma marketing teams, this delivers several important benefits.
Tracked feedback. Every comment is logged against a specific version of the document, with the reviewer's identity and a timestamp. Nothing is lost, and there's a clear record of what each reviewer raised.
Version control. Proofing environments manage versioning automatically. The current approved version is always visible, superseded versions are retained for audit purposes, and there's no risk of a reviewer inadvertently working on an outdated draft.
Audit history. The complete review history, including all comments, revisions, and sign-off actions, is available in the system. This supports both internal quality assurance and external regulatory requirements.
Secure collaboration. Access to content under review can be restricted to authorized individuals, with role-based permissions controlling who can comment, who can approve, and who can only view. This is especially important for pre-launch materials or content containing commercially sensitive information.
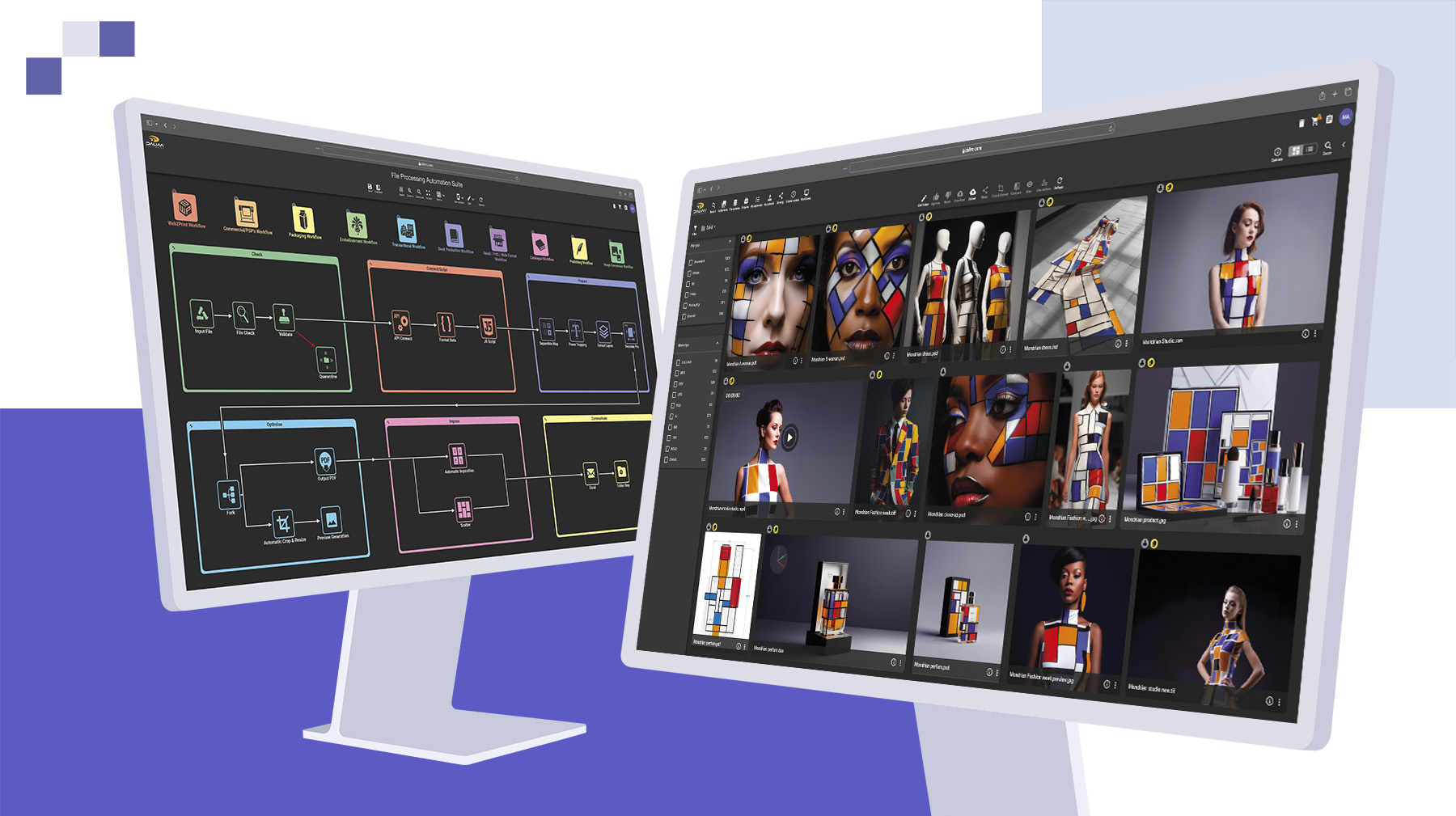
Platforms such as DALIM FUSION bring these capabilities together in an environment designed for complex, multi-stakeholder review workflows, including those that operate across multiple markets and languages.
Understanding the difference between a broken workflow and a functional one makes it easier to identify what needs to change.
| Inefficient Workflow | Compliant Approval Workflow | |
|---|---|---|
| Process | Ad hoc, varies by project | Standardized, documented stages |
| Communication | Email chains and file attachments | Centralized platform with tracked comments |
| Version control | Multiple versions circulating | Single source of truth, automatic versioning |
| Audit trail | Reconstructed manually | Automatically captured in the system |
| Visibility | Limited, requires status chasing | Real-time dashboards and status updates |
| Roles | Unclear, ad hoc involvement | Named owners at each stage |
| Review time | Unpredictable, often delayed | Defined timelines with escalation paths |
| Compliance assurance | Dependent on individual diligence | Built into the workflow structure |
The shift from the left column to the right isn't primarily a technology change. It's a process change, supported by technology.
Not all workflow tools are built for regulated environments. When evaluating solutions, pharma organizations should look for the following.
The system should capture a complete, tamper-evident record of all review actions, approvals, comments, and version history. This should be accessible for internal audits and available for regulatory review without requiring manual compilation.
Sensitive materials in review need to be protected. Look for role-based access controls, audit logs of who accessed what, and secure sharing mechanisms that don't rely on email attachments.
Pharma content comes in many forms, from promotional materials to patient support resources to HCP communications. The workflow system should be configurable to support different approval paths for different content types, rather than imposing a single rigid process.
Approval workflows don't exist in isolation. They need to connect with content management systems, digital asset management platforms, and the broader marketing operations stack. Integrations that reduce duplicate data entry and support end-to-end visibility are important in enterprise environments.
Pharma organizations often operate across multiple markets, business units, and languages. Approval workflow software needs to scale to accommodate global teams, localized content, and the complexity of managing multiple product lines and regulatory environments simultaneously.
DALIM's platform is built with these enterprise requirements in mind, supporting high-volume, multi-stakeholder workflows across international teams with the compliance infrastructure that regulated industries demand.
Improving an approval process is rarely a single intervention. It's a series of incremental steps that build toward a more structured, visible, and auditable system.
Map your current process before changing it. Understanding exactly how content currently moves from creation to approval, including all the informal workarounds and single points of failure, is essential before introducing new tools or processes. Process mapping often reveals problems that were assumed but never confirmed.
Reduce unnecessary stakeholders. Not everyone who is currently included in a review needs to be there. Working with compliance and regulatory leads to define who must review versus who is consulted can significantly reduce review cycle times without increasing risk.
Standardize workflows for each content type. Different types of content warrant different approval pathways. Defining these pathways clearly, documenting them in SOPs, and ensuring everyone understands them reduces the variation that causes delays and errors.
Introduce automation gradually. Wholesale process transformation is difficult to sustain. Starting with automation in one area, such as reviewer notifications and deadline reminders, and building out from there is more likely to succeed than attempting to automate everything at once.
Train teams and support adoption. New tools and processes only deliver value if people use them correctly and consistently. Investing in training, clear guidance, and ongoing support, particularly for reviewers who may only engage with the approval system periodically, is essential to making improvements stick.
Review and refine regularly. Approval workflows should be treated as living processes. Regular retrospectives to review where delays occurred, where compliance risks arose, and where feedback quality was low give teams the insight to keep improving.
A pharma marketing approval workflow is the structured process that governs how promotional and marketing materials are reviewed and signed off before publication. It typically involves sequential or parallel review stages across medical, regulatory, legal, and compliance teams, culminating in formal sign-off from a qualified signatory. The workflow exists to ensure that all materials are accurate, compliant with applicable regulations, and appropriately substantiated before they reach healthcare professionals, patients, or the public.
Pharma marketing approvals involve multiple stakeholders with different compliance responsibilities, strict regulatory requirements from bodies such as the MHRA, FDA, and EMA, and high consequences for errors. Unlike other industries, promotional claims must be medically accurate and fully substantiated. Any material that makes unsubstantiated claims, omits required safety information, or misrepresents a product can attract regulatory action. This level of scrutiny, while necessary, creates complexity that requires structured processes and appropriate infrastructure to manage efficiently.
The most effective approach combines process clarity, defined roles, and supporting technology. Mapping the current workflow, standardizing approval pathways by content type, clearly assigning ownership at each stage, and using centralized platforms for review and feedback all contribute to faster cycles without reducing rigor. Automation of notifications, deadlines, and version control removes administrative friction without removing human judgment from the critical review steps.
Purpose-built approval workflow platforms and online proofing tools provide the most robust support for regulated approval processes. Key capabilities to look for include centralized feedback, automatic versioning, tamper-evident audit trails, role-based access controls, configurable workflow stages, and scalability for enterprise teams. Platforms like DALIM FUSION are designed specifically for complex, multi-stakeholder creative workflows in regulated industries.
Online proofing replaces email-based document review with a structured digital environment where all feedback is captured against specific versions, every comment is attributed to a named reviewer with a timestamp, and the approval history is available for audit at any time. This eliminates the version confusion and undocumented feedback that create compliance risk in traditional review processes. It also makes it easier to manage concurrent reviews across multiple markets or languages.
An audit trail is a complete record of every action taken on a document during the approval process, including who reviewed it, what comments they made, what revisions were made, when each stage was completed, and who gave final sign-off. In regulated environments, audit trails are required to demonstrate that the correct review process was followed. Systems that capture this automatically, rather than requiring manual reconstruction, are considerably more reliable.
The primary risks are regulatory non-compliance, brand and reputational damage, and operational inefficiency. Non-compliant materials can lead to regulatory warnings, mandatory withdrawal of materials, and, in serious cases, legal consequences. Version control failures can result in unapproved content being published. Poor audit trails mean organizations can't demonstrate compliance if challenged. And slow, disorganized reviews delay campaigns and frustrate the teams responsible for delivering them.
Global approval workflows need to account for different regulatory requirements in different markets, multiple languages, local legal review, and time zone differences across review teams. Centralized platforms that support localized content workflows, allow market-specific review stages, and consolidate audit trails across regions are better equipped to handle this complexity than ad hoc email-based processes. Establishing clear global standards with flexibility for local market requirements is a governance principle that underpins effective global workflows.
Managing creative approvals in pharma marketing is genuinely difficult. The regulatory environment demands rigor. The business environment demands speed. And the two have to coexist.
The teams that do this well haven't found a shortcut around compliance. They have built structured, visible, well-supported workflows that make compliance the default rather than the afterthought. They have defined roles, standardized processes, removed unnecessary friction, and invested in infrastructure that captures the evidence they need and keeps content moving.
Technology is a significant part of this, but it's not the whole answer. The most capable approval workflow software won't fix a process that has no clear ownership or a team that hasn't agreed on how reviews should work. The tools enable the process. The process has to be designed intentionally.
For pharma marketing teams looking to reduce delays, manage compliance risk, and give their creative and review teams a better working environment, the starting point is the same: map what you have, identify where it breaks, and build toward something that works by design.
DALIM's workflow and online proofing capabilities are built to support exactly that kind of environment, providing the structure, visibility, and audit infrastructure that regulated marketing teams need to move efficiently without losing control.
Ready to see how DALIM supports compliant creative workflows in pharma and regulated industries? Get in touch with the team.

1 min read
Most marketing teams are busier than ever. Campaigns are running across more channels, content volumes are increasing, and the pressure to deliver...

1 min read
There's a moment most marketers know well. A campaign is nearly ready. The creative looks good, the copy is tight, and the deadline is tomorrow. Then...
1 min read
Why Your Print Shop’s Best Salesperson Might Be a DAM If you’re in the print business, the term “Digital Asset Management” or DAM probably brings a...